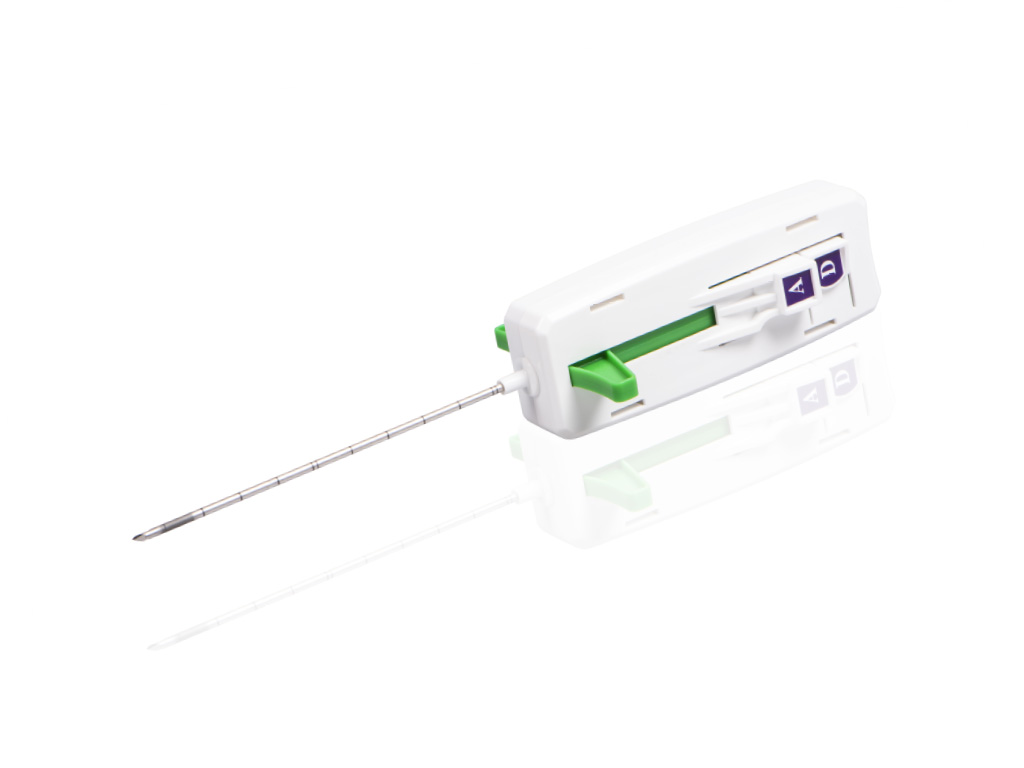
MEDAX is pleased to announce the April 2021 release of the MedOne Ultra, MedEasy, Med-L & Med-B for sales in the INDIA.
MedOne Ultra is programmable automatic disposable biopsy system ideal for both CT and Ultrasound guided percutaneous biopsy procedures.
Some key features of MedOne Ultra are:
- Automatic or sequential firing modes
- A wide selection of gauge sizes and lengths; 12-20 ga, 8-20 cm
- Packaged with or without a coaxial introducer needle set
MedEasy is semi-automatic disposable biopsy system ideal for both CT guided percutaneous biopsy procedures.
Some key features of MedEasy are:
- Light weight & ergonomic shape
- A wide selection of gauge sizes and lengths; 12-20 ga, 8-20 cm
- Packaged with or without a coaxial introducer needle set
- Coaxial set includes a ‘blunt tip’ stylet for atraumatic passage from the entry site to the biopsy site
Med-L is bone marrow aspiration and biopsy needle with special extraction cannula.
Some key features of Med-L are:
- Colour coded ergonomic handle
- Separate Extraction Cannula to harvest the specimen with minimal crush artifact
- A wide selection of gauge sizes and lengths; 8-13 ga, 10-15 cm
Medax Medical Devices India Private Limited
Registered Office: 602, Charkop, Mitashu Chs Ltd, R D P 6, Sector 6, Near TJSB Bank, Kandivali (W), Mumbai, 400067, Mumbai City, Maharashtra, India.
Warehouse: Shop No.1, Mahasagar Chs, Plot No. 112, Sector No.4, Opp Haryana Bhavan, Charkop,Kandivali West , Mumbai 400067, Maharashtra, India. - This email address is being protected from spambots. You need JavaScript enabled to view it.
MEDAX S.R.L. Unipersonale
Via Sandro Pertini, 4 - 41039 - San Possidonio (MO) - Italy
Company direct No. : +39 0535 1812757 This email address is being protected from spambots. You need JavaScript enabled to view it.
Vat N. /Fiscal Code N. Iscriz. Reg. Impr. 02669860369 - N. REA: MO 403036 - Capitale Sociale Euro 100.011,00 i.v.
In accordance with the requirements of the medical device 93/42/EEC directive and its relevant updates. All products undergo intensive clinical testing and are fully EC and FDA approved.